Click on an icon below to learn more:
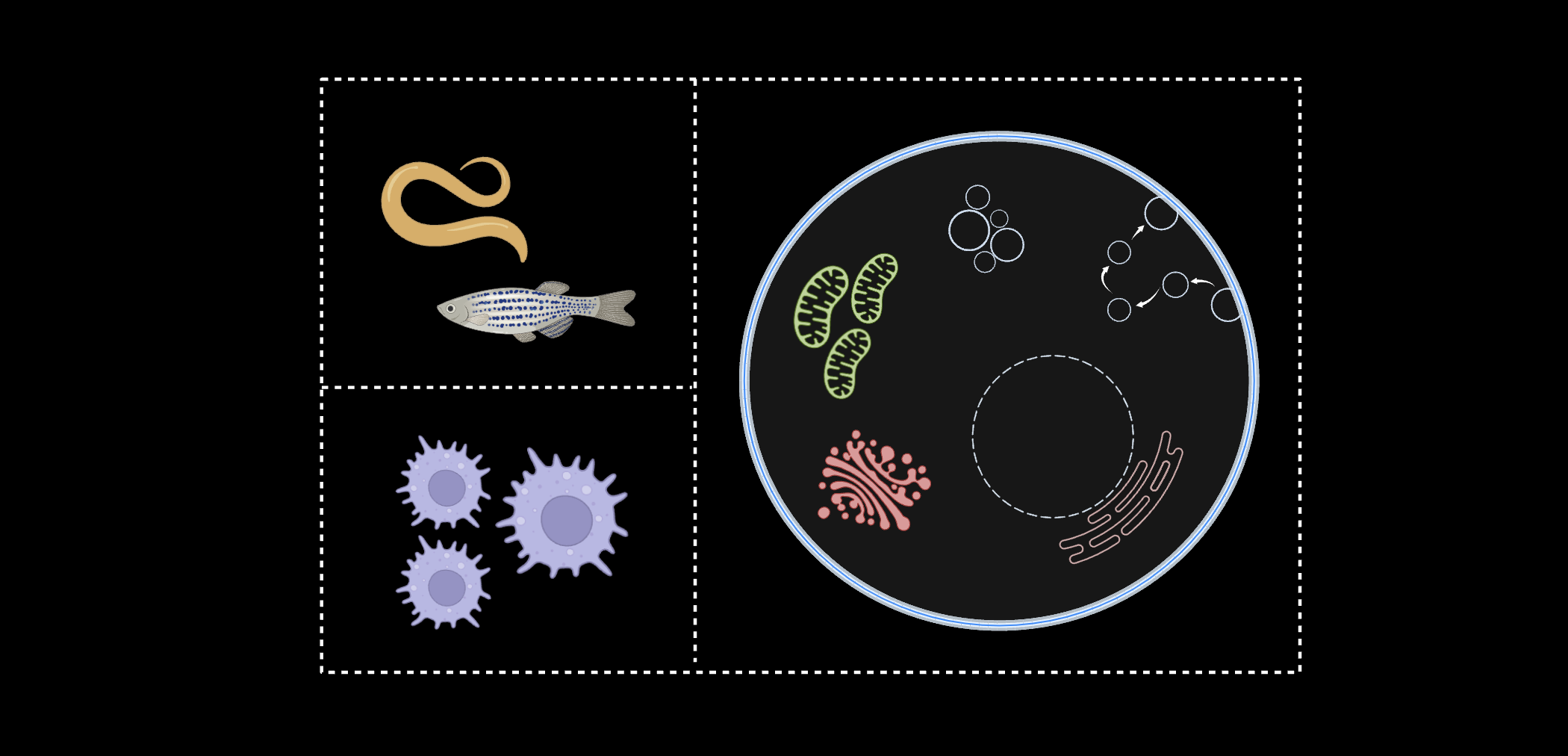
Mapping Organelles in vivo
With the use of C. elegans and zebrafish, we have been able to target our devices in a tissue-specific manner. For example, the development of new synthetic receptors in
C. elegans has allowed us to specifically target neuronal plasma membranes, endolysosomes of cholinergic or glutamatergic neurons, or lysosomes of intestinal epithelial cells. We also developed two new nanodevices for C. elegans chloride ion measurement: Clensor, which is pH-insensitive, and ChloropHore, which maps chloride and pH simultaneously.
With the use of NOckout in zebrafish, we were also able to quantify NOS2 activity in the phagosomes of zebrafish microglia.
Understanding Phagosomes
Phagosomes are the product of immune cell engulfment of foreign debris. We created a DNA nanodevice, z-cHOClate,
to measure pH and HOCl levels in the phagosomes of macrophages, monocytes, and neutrophils. We have also completed work in other areas of immunology, such as using DNA nanodevices to
deliver cathepsin inhibitors to the lysosomes of tumor-associated macrophages (TAMs).
The Trans-Golgi Network
The trans-Golgi network (TGN) contributes to the sorting, modifying, and tagging of proteins from the endoplasmic reticulum and is part of the retrograde pathway. We once again deployed the I-switch to measure pH in the TGN.
We have also used another nanodevice, NOckout, to measure NO levels and uncover the mechanisms of Nitric oxide synthase 3 (NOS3).
Recently, with the creation of pHlicKer, we have measured potassium levels within the TGN in a pH-independent manner.
Endoplasmic Reticulum
The endoplasmic reticulum (ER) is the primary manufacturing center of the cell. Our work seeks to further explore the
ionic contents of the ER and how they contribute to ER structure and function.
Endosomal Pathway
The endosomal pathway contains early endosomes, late endosomes, and lysosomes. It is a highly dynamic environment used for transporting cargo into various cellular compartments.
With the creation of the I-switch, our lab measured the pH of various compartments along the endosomal pathway to further understand how pH
is related to the maturation of endosomes within the cell. Sodium and potassium measurements using our nanodevices RatiNa and pHlicKer,
respectively, have further supported our understanding of endosomal maturation by mapping the ion levels of endosomes as they mature.
Lysosomes and Recycling Endosomes
Lysosomes are a key player in the endosomal pathway and are responsible for the breakdown of biological material within cellular environments.
Our work has led to the characterization of lysosomal lumenal pH and chloride, as well as reactive species such as hypochlorous acid (HOCL).
Recently, we have also uncovered the calcium levels of this organelle, as well as elucidating a mechanism for calcium entry into the lysosome with our nanodevice, CalipHlour.
In the recycling endosome, another step along the endosomal pathway, we have used our DNA nanodevices to quantify parameters such as pH, chloride, and membrane potential.
Mitochondria
There is still much to be learned from the "powerhouse" of the cell. With our innovative probes,
we are investigating the chemical and physical properties of this exciting organelle.
 Email:
Email: yamuna@uchicago.edu
2014 - Professor, University of Chicago
2014: Assoc Professor, National Centre for Biological Sciences (NCBS)
2009-13: Reader, NCBS, Bangalore
2005-9: Fellow, NCBS, Bangalore
2001-05:
1851 Research Fellowship, University of Cambridge, UK
2002: Ph. D., Indian Institute of Science (IISc), Bangalore,
1997: M.S., IISc, Bangalore,
1993: B. Sc. Madras University
Our lab loves exploring the rich chemistry within the living cell’s myriad reaction vessels – its organelles. How does the lumenal chemical composition of an organelle – that has been optimized over evolutionary timescales – drive the biochemistry that occurs within to impact organelle function, thereby cell function, then tissue function and finally, organism physiology? To answer this, we build quantitative chemical maps of organelle lumens using the tools of bionanotechnology. Evolution has produced an overwhelming number and variety of biological devices that function at the nanoscale or molecular level. Bionanotechnology exploits our understanding of cellular machinery to develop solutions to problems in science and engineering. Our research exploits nucleic acid structure and dynamics to create DNA-based nanodevices for quantitative chemical imaging of living systems.


